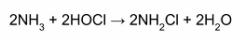Chlorine
|
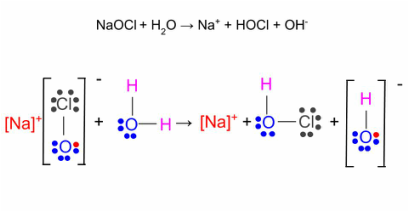
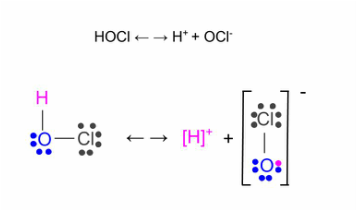
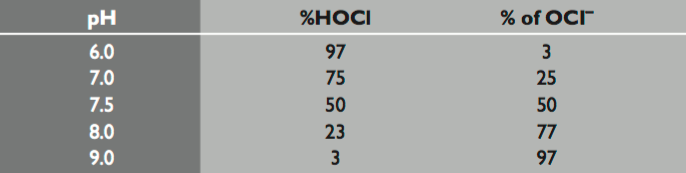
Chlorine is the most effect and commonly used sanitizer in swimming pools. Chlorine's role is to immediately react with any containments that enter the pool. The most desirable and effective form of chlorine is known as free chlorine. Free chlorine consists of hypochlorous acid (HOCl) and an hypochlorite ion. In order to obtain free chlorine in the pool water, one must add some form of chlorine (see Image 6 for different types of chlorine available). For this example, we'll assume its sodium hypochlorite (NaOCl). When the NaOCl enters the pool it reacts with the water, resulting in free chlorine (see Diagram 1 for the chemical equation). In the actual chemical reaction, only HOCl is formed, however HOCl easily dissociates into the hypochlorite ion (see Diagram 2). Therefore, together, the two exist in an equilibrium. This means that if 25% of the free chlorine present in the pool is hypochlorous acid then the other 75% is hypochlorite and visa versa. The percent of how much one or the other is actually relies on the pH levels (see Image 7 for specifics). The hypochlorous acid is the part of free chlorine that is responsible for sanitation. Therefore, it would seem logical to lower the pH so that there was as much as possible. However, as earlier mentioned, chlorine is ineffective at low levels of pH.
“Free chlorine is highly reactive and once added to water, quickly attacks bacteria as well as bather and other wastes” (27). Most of the waste that enters the pool contains ammonia (NH3). Below is a chemical reaction of HOCl reacting with ammonia. Once the HOCl has reacted with ammonia, it is no longer known as free chlorine. Instead, it is referred to as combined chlorine or chloramine. While combined chlorine is stable, it's "a very poor sanitizer, it is the agent responsible for eye burn and skin irritation and results in the unpleasant chlorine odor often referred to as a pool with 'too much chlorine'" (29). Therefore, it is only recommended to have no more than 0.2 ppm in the pool at a time. However, if enough free chlorine is present, the combined chlorine won't last long. The final step in the sanitation process is for any additional free chlorine to further break down the combined chlorine into hydrogen, nitrogen gas, and liquid water (30). Every time this process occurs, it depletes the amount of free chlorine available to react with future waste. Therefore, the chlorine levels must be carefully monitored and adjusted. Thus, if the chlorine is kept at proper levels, it is extremely effective at keeping the pool safe and clean. (31)
|
|