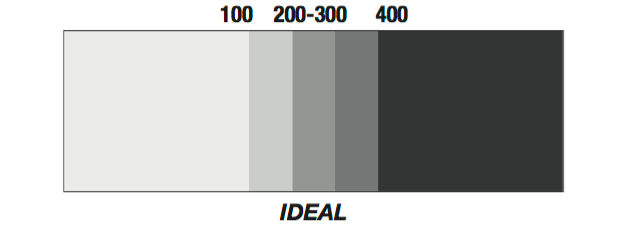
Calcium Hardness
Calcium hardness is the total amount of calcium dissolved in the pool water. Negative effects occur if the calcium level is too high or too low. Therefore, calcium hardness must be kept in-between 100 and 400 ppm (see Image 3). If calcium hardness goes below 100 ppm, the water will seek calcium elsewhere, typically from the metal fittings and plaster in the pool, causing it to corrode. If the calcium hardness is too high, it becomes unstable (which will be explained later). Usually, the calcium is soluble in the water, however, some aspects can cause it to precipitate (cause (a substance) to be separated in solid form from a solution (18)). If there is too much calcium, the pH or temperature is too high, or total alkalinity is unbalanced the calcium becomes unstable. If this happens, the water will become cloudy, as the precipitated calcium becomes visible. If this occurs, a sequestering agent can be added which will strengthen the bonds between the pool water and calcium so the calcium won't precipitate. (19)